Bactochem Plant Capacity:
360,000 Mega
Heparin Sodium

Heparin Sodium (IP/BP/USP)
Heparin is a heterogenous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans, possessing anticoagulant properties. It is composed of polymers of alternating derivations of α-D-glucosamido (N-sulfated O-sulfated or N-acetylated) and O-sulfated uronic acid (α-L-iduronic acid or β-D-glucuronic acid).
Heparin Sodium API Manufacturers is a sterile preparation of heparin sodium derived from porcine intestinal tissue, standardized for anticoagulant activity, in water for injection. It is intended for intravenous or deep subcutaneous administration. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Grade:
Injectable/Topical/Crude
Specification:
BP/EP/USP/IP
Heparin Sodium Injection is indicated for:
- Prophylaxis and treatment of venous thrombosis and pulmonary embolism;
- Prophylaxis and treatment of thromboembolic complications associated with atrial fibrillation;
- Treatment of acute and chronic consumption coagulopathies (disseminated intravascular coagulation);
- Prevention of clotting in arterial and cardiac surgery;
- Prophylaxis and treatment of peripheral arterial embolism;
- Anticoagulant use in blood transfusions, extracorporeal circulation, and dialysis procedures.
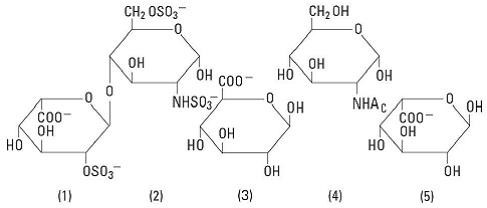
Structure of heparin sodium
